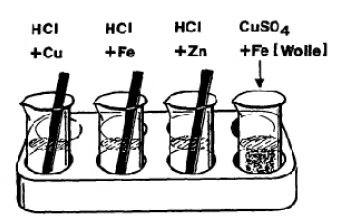
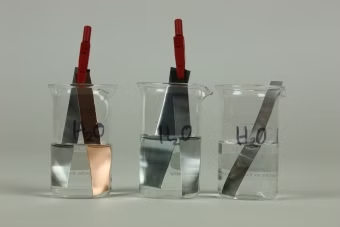Principle
Items made of iron, even if they are much thicker and mightier than gold and silver items, vanish within a few decades in air, water or earth. Such destruction of metals is summed up together under the term "corrosion" (from the Latin corrodo). Electro-chemistry enables an explanation to be found for such processes and also points out ways of protecting valuable metal items and technical installations. To observe some corrosion processes in acids, salt solutions and water, and gain knowledge on protection against corrosion.
Learning objectives
- Principle of metal corrosion
Benefits
- Easy teaching and efficient learning by using interactive experimentation PHYWE-Software
- Experiment is part of a complete solution set with experiments for the topic Electrochemistry matched with international curriculum: all topics are covered